Resources
This page is a central hub for Norn Group's research outputs, tools, and written analyses.
Aimed at longevity researchers, biotech founders, funders, and anyone working to extend healthy human lifespan.

An overview of the endeavor of extending healthy life, mapping the path from basic research to therapies and highlighting key bottlenecks slowing progress.
It covers the people, funding, tools, trials, and to-be solved bottlenecks that turn aging research into real therapies and maps how to scale the field for far greater impact.
An in-depth interactive exploration of the system for extending healthy life, tracing how research becomes therapies and where key bottlenecks and opportunities shape progress.
Clicking through the various stages reveal circular and text nodes containing in depth information about the progress in the field.
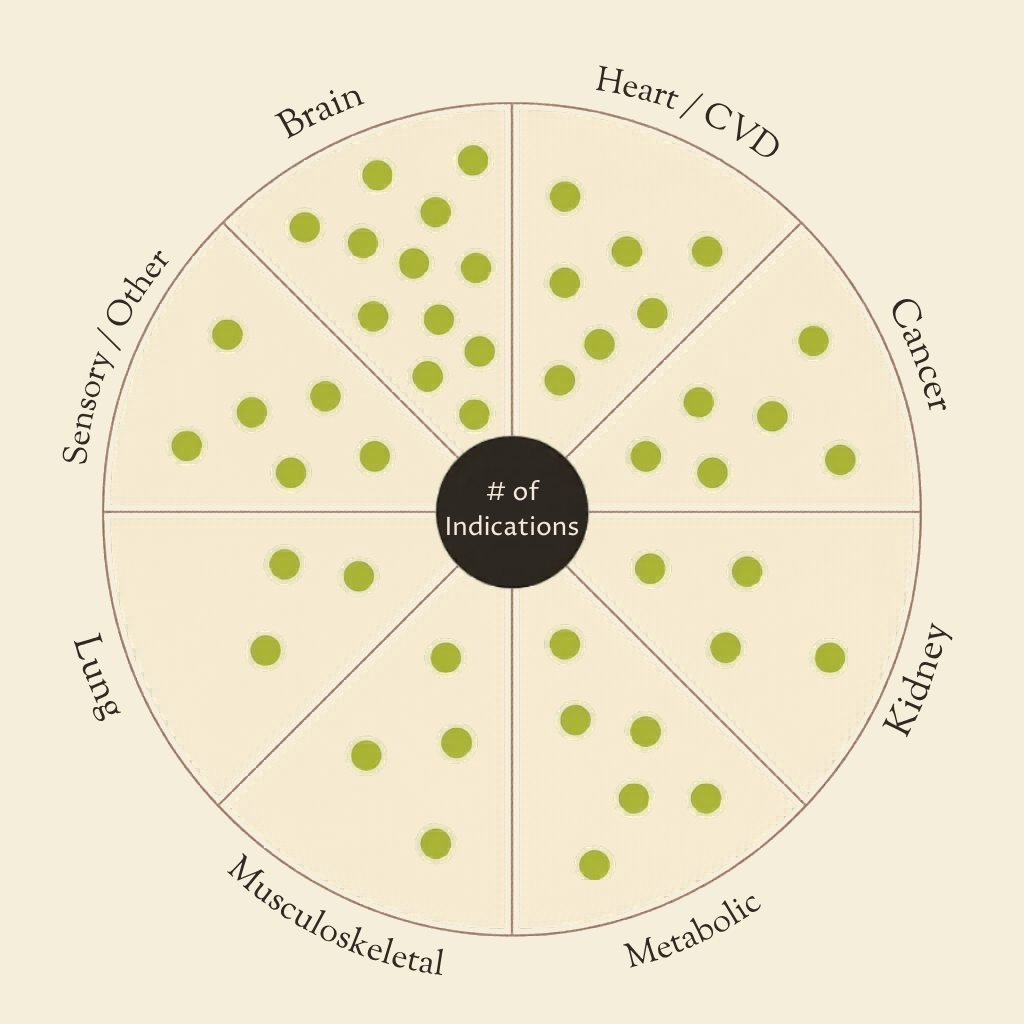
A spreadsheet overview of major age-related diseases, created as a resource to help longevity startups and aging researchers to identify opportunities for viable drug programs beyond lifespan studies.
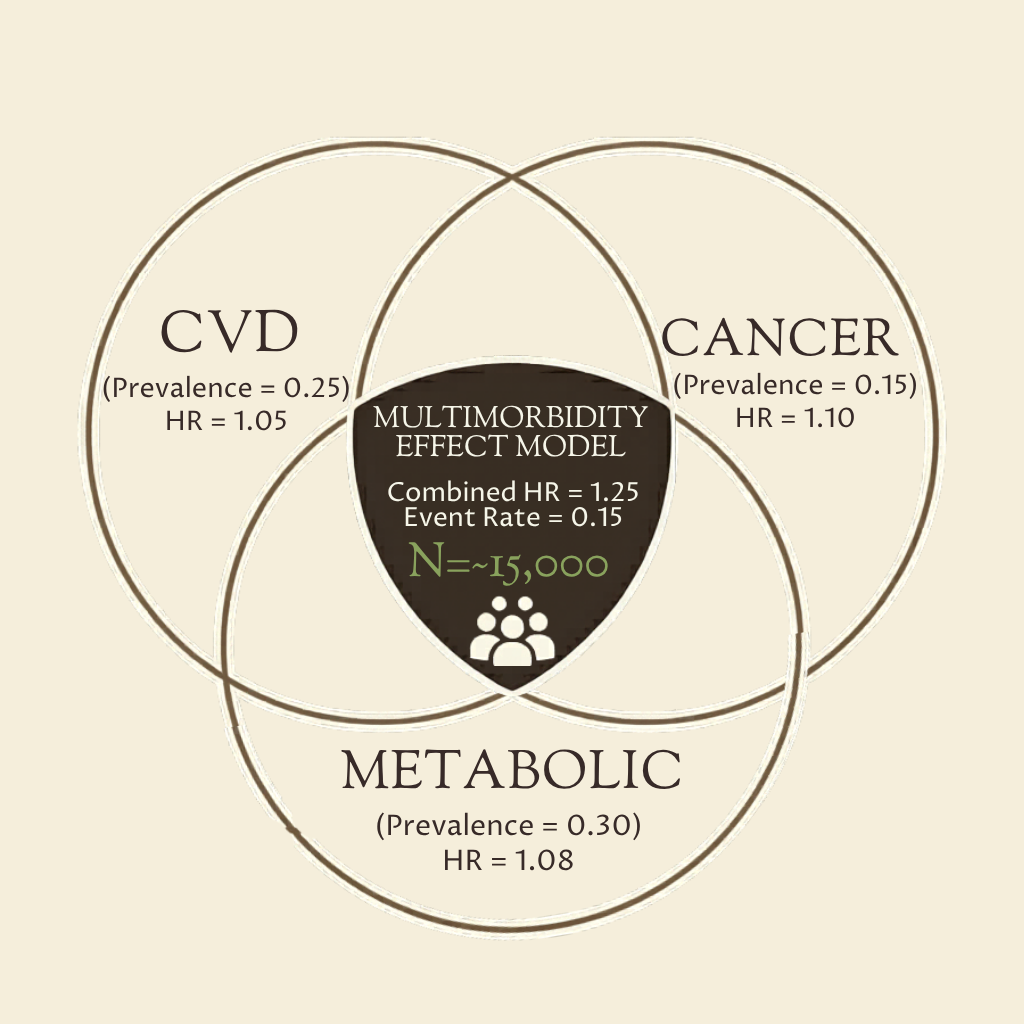
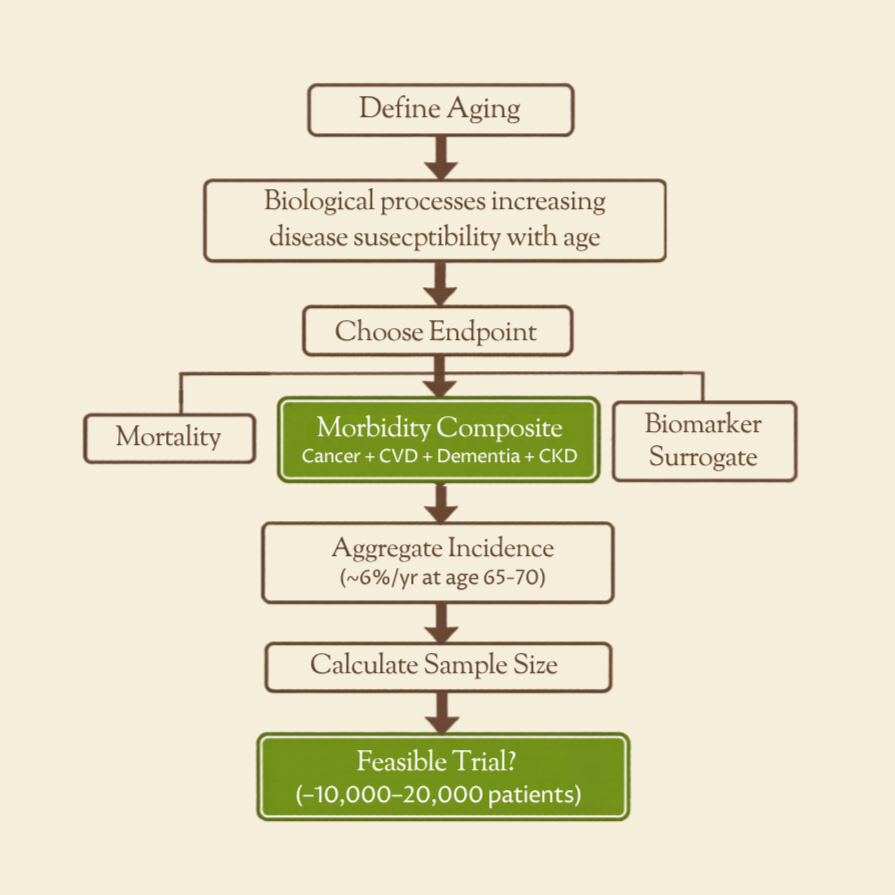
From our piece: “How to Design a Clinical Trial for Aging” we produced this tool for estimating the sample size and feasibility of aging-prevention trials that use a composite endpoint across several age-related diseases.
Users can input hazard ratios for expected effects on cancer, cardiovascular disease, dementia, and late-stage CKD; the calculator then estimates a composite hazard ratio and trial size.
Its purpose is to show whether a broad aging trial looks economically viable. The model assumes 65–70 population-average incidence and independence between diseases, so results are approximate rather than definitive.

Writing
These pieces from Norn Group’s newsletter include substantive research outputs, including comprehensive analyses of bottlenecks, practical frameworks, and tools designed for repeat use.
Sign up for Longevity Industrialized with your email address and get our latest pieces, programs’ news and other updates straight to your inbox.
A piece produced by Nicolas di Leo, as a part of our Talent Bridge Program.
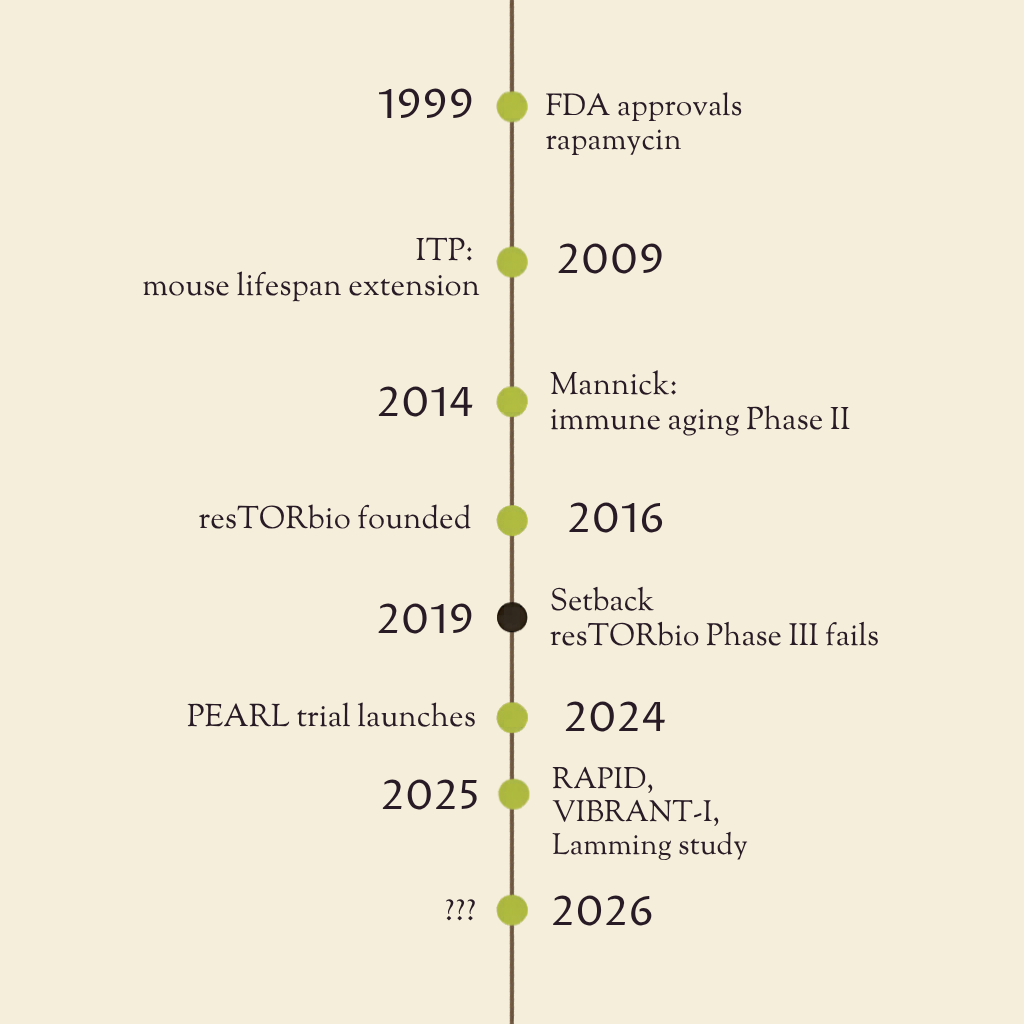
An overview of what mTOR-targeting interventions can teach longevity trial design, with a focus on rapamycin and related programs. It reviews past and ongoing human studies, highlights lessons from biotech efforts, and examines the practical challenges of endpoints, safety, biomarkers, and indication selection in translating geroscience into clinically actionable trials.
A practical guide to designing a pivotal clinical trial for aging, focused on endpoints that could support real-world approval and reimbursement. It explores composite morbidity and mortality endpoints across major age-related diseases, compares trial strategies, and includes a calculator for estimating sample size and feasibility.
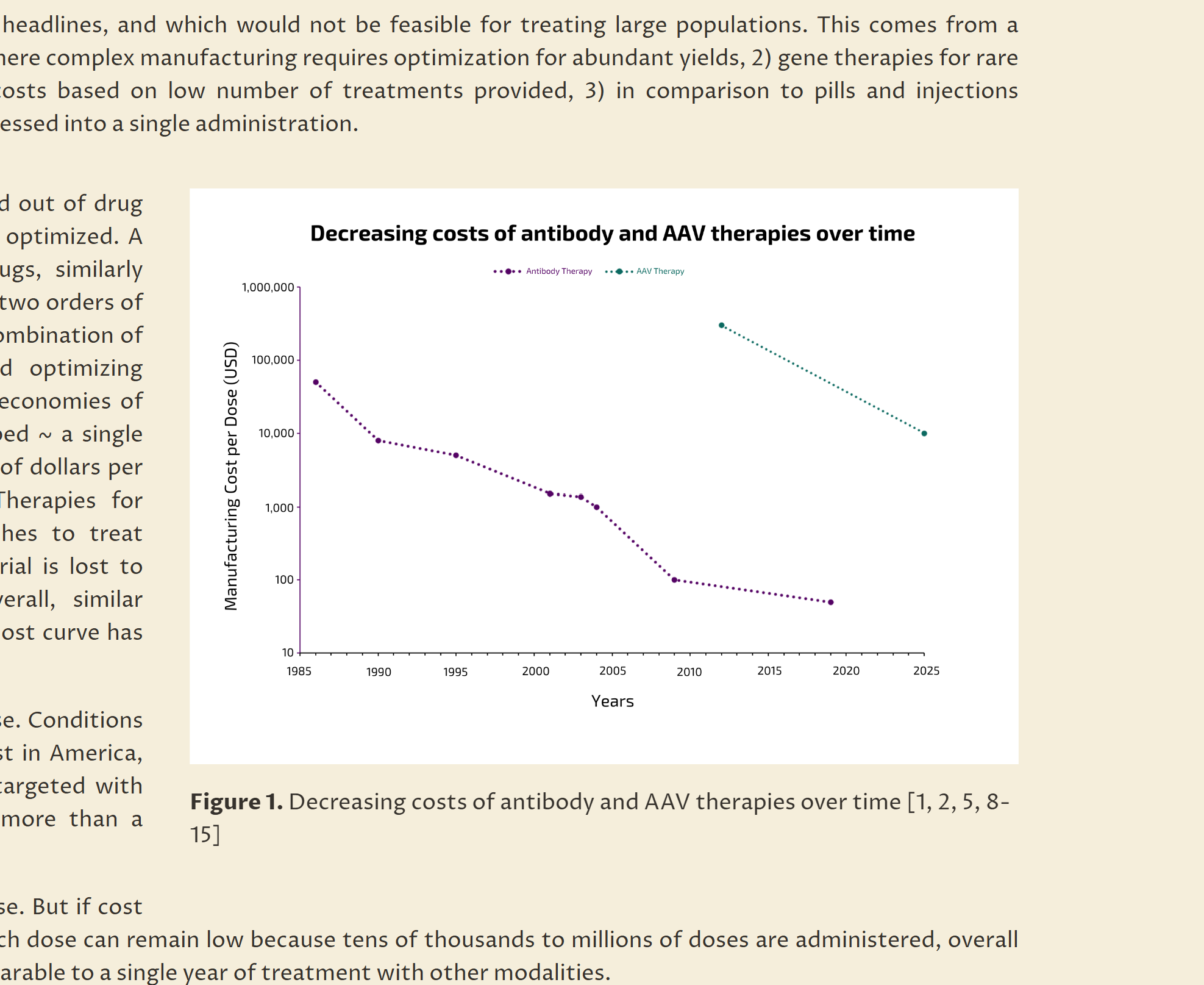
An overview of how AAV gene therapy could expand beyond rare monogenic disorders into common diseases. It examines the economics, safety, eligibility, durability, and manufacturing considerations that shape this transition, and explores where gene therapy may offer practical advantages over conventional drug modalities.
A piece produced by Marton Meszaros, as a part of our Talent Bridge Program.
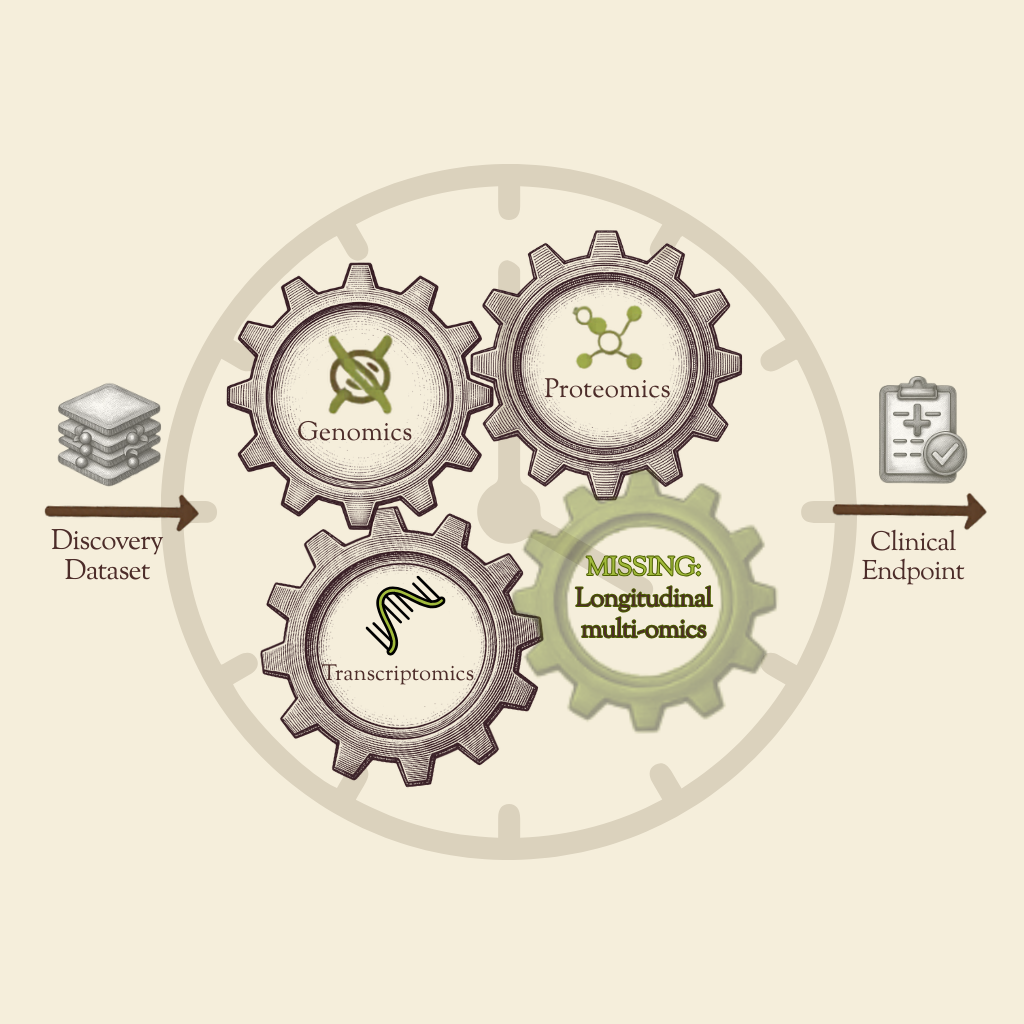
An overview of why aging biomarker discovery is constrained by the human datasets it depends on. It examines what useful aging biomarkers must achieve in drug development, where current cohorts fall short, and what new data resources may be needed to support clinically relevant biomarker discovery and validation.

Looking for more resources?
See the Big Picture - Deep Dive where we have a in-depth mapping of external resources and initiatives in the field.