Advancing Longevity Trials: Lessons from mTOR-Targeting Interventions
By Nicolas Di Leo
The mechanistic target of rapamycin (mTOR) pathway is central to cell growth and aging, integrating signals from nutrients and stress to regulate metabolism, protein synthesis, and autophagy. Decades of research across various species have identified mTOR inhibition as a powerful strategy for extending lifespan and healthspan. In particular, the mTORC1 inhibitor rapamycin has repeatedly been shown to delay aging in yeast, worms, flies, and mice even when treatment is started late in life [1,2]. These findings position mTOR as one of the most validated targets for translating longevity interventions to humans. Yet turning this laboratory success into clinical practice has proven challenging.
Early human trials of mTOR inhibitors hint at improved immune function, cardiovascular markers, and skin health with little side effects [3]. However, they also expose critical gaps: how to measure “aging” in trials, how to balance efficacy with safety in otherwise healthy people, and how to design studies that regulators and industry can get behind.
This report reviews the clinical translation of mTORC1-targeting interventions for aging and age-related disease, with a focus on rapamycin and related strategies outside of traditional immunosuppressive use. It begins with a brief biological background on mTOR and rapamycin, then outlines why mTORC1 remains one of the most compelling pathways for geroscience translation across major domains of age-related pathology. The report next examines past human clinical trials, followed by a review of ongoing and emerging studies, enriched by exclusive insights from the principal investigators leading these programs. It then synthesizes the main translational gaps that continue to limit progress, including endpoint selection, biomarker validation, safety monitoring, and indication prioritization, and discusses practical directions for improving future trial design. The overall aim is to provide a structured, evidence-based assessment of where the field stands, what has been learned from prior efforts, and what is needed to make mTORC1-targeting therapies more interpretable, testable, and clinically actionable in geroscience and longevity medicine.
mTOR and Rapamycin: A Brief Background
mTOR is a conserved serine/threonine kinase that forms two complexes, mTORC1 and mTORC2, which regulate cell growth and metabolism in response to nutrients, growth factors, and energy status. mTORC1, in particular, promotes anabolic processes (protein/lipid synthesis) and inhibits autophagy. Inhibition of mTORC1 triggers a shift from growth to maintenance: boosting autophagy, improving proteostasis, and mitigating cellular senescence [1]. This mechanism helps explain why chronic dietary restriction, which suppresses mTOR signaling, extends lifespan across many species. Rapamycin, a natural product discovered from Streptomyces bacteria, pharmacologically recapitulates key aspects of this low-nutrient state by binding FKBP12 and preferentially inhibiting mTORC1. Importantly, rapamycin remains one of the most reproducible longevity interventions identified to date: it was the first small molecule shown to robustly extend lifespan in mammals, and it continues to stand out from most candidate “anti-aging” compounds in the strength and consistency of the evidence. In 2009, Harrison et al. from the NIA Intervention Testing Program (a multi-center platform using genetically heterogeneous, outbred mice) demonstrated that rapamycin administration starting as late as 20 months of age increased median lifespan by ~10–16% [4]. Subsequent studies have replicated lifespan extension across multiple cohorts, strains, and both sexes, supporting the conclusion that mTORC1 is not a niche target but a central regulator of mammalian aging biology.
Encouragingly, genetic evidence aligns with pharmacology: mice or flies with reduced mTORC1 activity (or increased upstream inhibitors like TSC) tend to live longer, whereas hyperactive mTOR signaling accelerates aging phenotypes [1]. Meanwhile, chronic hyperactivation of mTOR is implicated in many age-related diseases (e.g. cancer, type 2 diabetes, neurodegeneration) [1]. These insights made mTOR an attractive target to slow aging and prevent multiple chronic diseases at once. Rapamycin and its analogs (“rapalogs” such as everolimus) are already FDA-approved for other indications, including organ transplant rejection (as immunosuppressants), certain cancers, and rare disorders like tuberous sclerosis complex (TSC) [1]. Could lower doses or intermittent schedules of these same drugs improve healthspan in otherwise healthy older adults? This provocative idea has catalyzed numerous trials in the past decade.
However, translating an “anti-aging” therapy into the clinic has proved to be tricky. Aging itself is not recognized as a disease by regulators, so trials must target specific age-related conditions or risk factors. Furthermore, measuring aging is non-trivial – unlike a single disease, aging manifests as a multifaceted decline. Safety is another paramount concern: rapamycin at therapeutic doses can cause immunosuppression, metabolic disruptions, mouth ulcers, and other side effects [1]. For “privileged” use in generally healthy populations, even mild toxicities need to be minimized. Thus, a central challenge has been finding dosing strategies that inhibit mTORC1 enough to slow aging, while avoiding excessive mTORC2 inhibition that may underlie many side effects [2].
In summary, the rationale for targeting mTOR in longevity is compelling: it’s a conserved aging pathway with pharmacological tools already in hand. But the path forward requires careful trial design to demonstrate benefits in humans without unacceptable risks. Below we review what has been learned so far from preclinical studies and human trials, and how this informs current efforts to harness mTOR inhibition to combat chronic diseases of aging.
mTOR’s Clinical and Translational Appeal
A consistent theme in longevity research is that, to gain traction clinically in 2026, given the bureaucratic hurdles in place, therapies should target well-defined chronic diseases or risk factors, thereby treating “ageing” by treating an age-related condition. mTOR inhibitors exemplify this approach. Rather than proposing rapamycin as a general anti-ageing drug (an idea that faces regulatory hurdles), many researchers are testing rapamycin in specific diseases of aging, with the hope that success in those domains will validate its broader geroprotective effects. This section highlights some chronic disease contexts where mTOR targeting is being pursued as a longevity strategy, and what has been learned so far.
In immune senescence, aging is characterized by reduced naïve T-cell function, impaired vaccine responsiveness, weaker antiviral defenses, and a chronic inflammatory baseline that paradoxically coexists with immunodeficiency. mTOR activity is a central regulator of immune-cell activation, differentiation, and memory formation, and excessive or dysregulated signaling with age can contribute to maladaptive immune states. In preclinical systems, partial mTORC1 inhibition often improves aspects of immune function and reduces inflammatory tone, and in humans the most consistent early clinical signals for mTOR modulation have come from immune-related endpoints and infection susceptibility, supporting the idea that immune aging is one of the most tractable entry points for geroprotective translation.
Cancer provides a second, compelling rationale because mTORC1 is a core effector of growth and proliferation downstream of oncogenic pathways, and many tumors exploit mTOR signaling to sustain biomass accumulation, survival, and metabolic reprogramming. Aging increases cancer risk through accumulated mutations, epigenetic drift, and changes in the tissue environment that favor clonal expansion. In preclinical models, dialing down mTORC1 tends to reduce tumor initiation and progression in multiple settings, partly by limiting proliferative signaling and partly by improving cellular housekeeping and reducing inflammatory, pro-tumor microenvironments. Clinically, mTOR inhibitors are already established in oncology and in genetic disorders characterized by hyperactive mTOR signaling, demonstrating that the pathway is druggable in humans. For geroscience, the translational opportunity is not necessarily to treat advanced cancers with low-dose mTOR inhibition, but to test whether carefully dosed, long-term modulation can reduce cancer-promoting conditions that build up with age, potentially delaying incidence or slowing progression of early lesions.
Cardiovascular aging is increasingly understood as a convergence of endothelial dysfunction, vascular stiffness, chronic inflammation, impaired autophagy, mitochondrial decline, and maladaptive remodeling of heart tissue. mTORC1 influences all of these processes, including smooth muscle cell growth, fibrosis pathways, inflammatory signaling, and cellular quality control. In animal models, partial mTORC1 inhibition can improve aspects of cardiac function, reduce age-associated hypertrophy and fibrosis signals, and support better vascular function.
Metabolic disease sits in a similar category. mTOR integrates nutrient availability with anabolic signaling, and chronic overnutrition can lock tissues into a persistent growth mode that contributes to insulin resistance, ectopic lipid deposition, and impaired metabolic flexibility. In preclinical systems, modulating mTORC1 can improve metabolic resilience in some contexts by reducing inflammatory signaling and enhancing autophagy, but sustained inhibition that spills into mTORC2 can impair insulin signaling and glucose control. Human experience mirrors this complexity: metabolic effects depend strongly on dose, schedule, baseline metabolic state, and duration.
Neurodegeneration is another domain where the biology points to mTOR as a plausible upstream lever. Many neurodegenerative diseases involve failures of proteostasis, impaired clearance of misfolded proteins, mitochondrial dysfunction, and chronic neuroinflammation, all processes that mTORC1 can influence through its control of autophagy and cellular stress responses. In preclinical models, reducing mTORC1 signaling often improves clearance pathways and can blunt inflammatory and synaptic dysfunction phenotypes in models of protein aggregation. Translationally, the challenge is that the brain is both difficult to access for pharmacodynamic readouts and sensitive to perturbations in protein synthesis and synaptic plasticity, which may render the therapeutic window impractical. Still, the conceptual appeal is strong: a pathway that enhances cellular clearance may be especially relevant in conditions in which accumulated cellular debris drive progressive loss of function.
Across these domains, the broader point is that mTOR is one of the few targets with a coherent mechanistic link between multiple age-related pathologies, and with real-world drug precedents that reduce risk.
Past Clinical Trials
Across human research to date, mTOR inhibition has been tested in several clinical contexts that go beyond immunosuppression, with the overarching goal of determining whether modulating this pathway can produce measurable improvements in age-related physiology. In general, these trials have used low or intermittent dosing and have focused on outcomes that can shift within weeks to months, including vaccine responsiveness and infection susceptibility, markers of systemic inflammation, cardiometabolic readouts, skin and tissue aging features, and functional measures such as strength, endurance, and aerobic capacity. A consistent theme is that the interventions are usually feasible and generally well tolerated in older adults, while also highlighting substantial heterogeneity in biological response across individuals, sex and endpoints. Collectively, the studies highlighted in the table below support the concept that mTOR modulation can influence human aging-relevant biology in multiple organ systems, but they also emphasize the importance of selecting appropriate populations, defining clinically interpretable endpoints, and incorporating mechanistic biomarkers that can confirm target engagement and help explain variability in outcomes.
| Name of first author and year of publication | Title of the study | Trial Phase | Number of participants and demographic | Drug/Intervention | Treatment Duration | Primary Outcomes | Key Findings & Safety Results |
|---|---|---|---|---|---|---|---|
| Joan B. Mannick et al. 2014 [5] | mTOR inhibition improves immune function in the elderly | Phase 2 | 218 volunteers aged 65 years and older without unstable underlying medical diseases (generally healthy). | Everolimus (RAD001) at doses of 0.5 mg daily, 5 mg weekly, or 20 mg weekly, compared to placebo. | 6 weeks of drug treatment followed by a 2-week drug-free interval prior to vaccination. | Enhancement of immune function as assessed by the serologic response (Hemagglutination Inhibition titers) to seasonal influenza vaccination. | Low-dose Everolimus (0.5 mg daily or 5 mg weekly) significantly enhanced the response to influenza vaccine by approximately 20% and reduced the percentage of exhausted PD-1-positive T cells. It also broadened the response to heterologous influenza strains. The drug was relatively well tolerated; the most common adverse event was mouth ulcers (stomatitis), which were mostly mild. One severe adverse event (mouth ulcers) was reported in the 20 mg weekly group. |
| Joan B. Mannick et al. 2018 [6] | TORC1 inhibition enhances immune function and reduces infections in the elderly | Phase 2a | 264 elderly volunteers aged 65 years and older, without unstable underlying medical diseases (generally healthy). | Interventions included 0.5 mg Everolimus (RAD001) once daily, 0.1 mg Everolimus once daily, 10 mg Dactolisib (BEZ235) once daily, or a combination of 0.1 mg Everolimus and 10 mg Dactolisib once daily, versus placebo. | 6 weeks of drug treatment, followed by a 2-week drug-free interval and 9 months of follow-up. | Immune function as assessed by serologic response to influenza vaccination (hemagglutination inhibition geometric mean titer ratio) and reduction in the rate of infections. | The combination of Everolimus and Dactolisib met the primary endpoint with a greater than 20% increase in antibody titers for all three influenza strains. There was a significant decrease in the annualized rate of all infections (p=0.001) and respiratory tract infections (p=0.01). Treatments were well-tolerated with no deaths; diarrhea was the most frequent mild adverse event. Notably, rates of hyperglycemia and hypercholesterolemia were lower in treatment groups than placebo. Treatment also up-regulated interferon-induced antiviral gene expression. |
| Ellen Kraig et al. 2018 [7] | A Randomized Control Trial to Establish the Feasibility and Safety of Rapamycin Treatment in an Older Human Cohort: Immunological, Physical Performance, and Cognitive Effects | Phase 1 and Phase 2 (Pilot studies) | 25 generally healthy older adults (aged 70–95 years; mean age 80.5) with stable medical conditions. Phase 1 included males 80–95 years; Phase 2 included males and females 70–93 years. | 1 mg Rapamycin (Sirolimus) daily versus placebo. | 8 weeks to 4 months. | Safety and tolerability (basic metabolism via complete blood count, comprehensive metabolic panel, hemoglobin A1c, and oral glucose tolerance tests), cognitive function, physical performance, and immune aging parameters. | Rapamycin was well-tolerated with no significant changes in clinical laboratory, cognitive, or physical performance measures. No increases in blood glucose or insulin sensitivity were found. Statistically significant decrements were observed in erythrocyte parameters (hemoglobin, hematocrit, red blood cell count, mean corpuscular volume, and mean corpuscular hemoglobin), though they were not clinically significant. Adverse events included facial rash, stomatitis, and gastrointestinal issues. |
| Christina Lee Chung et al. 2019 [8] | Topical rapamycin reduces markers of senescence and aging in human skin: an exploratory, prospective, randomized trial | Exploratory Phase 1/2 | 36 participants (17 completed) greater than 40 years of age with photoaging and dermal volume loss, without major morbidities. | Topical Rapamycin cream (10 µM) applied to the dorsal side of one hand vs. placebo on the other. | 6 to 8 months. | Level of p16INK4A protein (a marker of cellular senescence) in the skin. | Significant reduction in p16INK4A protein (p=0.008) and increase in collagen VII (p=0.0077). Clinical improvement in skin appearance (fewer wrinkles, better tone) was noted. No treatment-related adverse events were reported; systemic absorption of rapamycin was not detectable in blood (detection limit <1 ng/ml). |
| Mitzi M. Gonzales et al. 2025 [9] | Rapamycin treatment for Alzheimer’s disease and related dementias: a pilot phase 1 clinical trial | Phase 1 (Pilot, open-label) | 10 participants aged 55–85 years (mean age 74) with mild cognitive impairment or early-stage dementia due to Alzheimer’s disease. | 1 mg Rapamycin (Sirolimus) per day. | 8 weeks. | Central nervous system penetrance (detection of drug levels in cerebrospinal fluid). | Rapamycin was not detectable in the cerebrospinal fluid. Twenty adverse events occurred, mostly mild (urinary urgency and diarrhea). Systolic blood pressure and hemoglobin A1c significantly increased, while erythrocyte parameters decreased. Significant increases were seen in cerebrospinal fluid biomarkers including phosphorylated tau-181, glial fibrillary acidic protein, and neurofilament light. No significant changes in cognition were found. |
| Mauricio Moel et al. 2025 [10] | Influence of rapamycin on safety and healthspan metrics after one year: PEARL trial results | Phase 2 | 114 healthy, normative-aging adults (aged 50–85 years) recruited online. | Compounded Rapamycin (5 mg or 10 mg weekly) versus placebo. | 48 weeks. | Visceral adiposity change (via dual-energy X-ray absorptiometry scan). | No significant change in the primary endpoint of visceral adiposity. Lean tissue mass and self-reported pain significantly improved in women using 10 mg weekly. General health and emotional well-being improved in the 5 mg group. Adverse events were similar across groups, though gastrointestinal symptoms were slightly more frequent in rapamycin users. One case of anemia in the 5 mg group resolved with transfusion. Compounded rapamycin had roughly one-third the bioavailability of commercial formulations. |
| Alexander J. Moody et al. 2025 [11] | Short-term mTOR inhibition by rapamycin improves cardiac and endothelial function in older men: a proof-of concept pilot study | Pilot (Proof of concept) | 6 community-dwelling men (aged 70–76 years) in good health with no known cardiac disease. | 1 mg Rapamycin (Sirolimus) per day. | 8 weeks. | Cardiovascular function (cardiac MRI) and endothelial function (laser-Doppler flowmetry). | Statistically significant improvements were observed in transmitral blood flow, peak flow rate, and maximal blood acceleration (diastolic function). Endothelial function (nitric oxide-mediated vasodilation) also significantly improved (p=0.020). End-systolic volume increased significantly. Safety labs showed mild declines in white blood cell count, mean corpuscular volume, and mean corpuscular hemoglobin, plus increases in hemoglobin A1c and LDL, all within normal clinical ranges. No adverse events were reported. |
| Jonas E. Svensson et al. 2024 [12] | Evaluation of rapamycin as a neuroprotective treatment in Alzheimer’s disease: a six-month single-arm open-label clinical pilot trial | Phase IIa | 14 participants 55-80 years with MCI or mild dementia | 7mg weekly rapamycin (Tablet Rapamune®) | 6 months (26 weeks) | Change in cerebral glucose uptake measured using [18F]FDG PET | Rapamycin was well tolerated. No significant metabolic or perfusion changes were observed in primary regions. Exploratory analyses showed increases in [18F]FDG SUVR in the putamen, insula, and anterior cingulate cortex, and thalamic CBF. Higher rapamycin blood concentrations correlated with increased [18F]FDG SUVR in several regions, including the temporoparietal cortex. CSF analysis demonstrated significant increases in total tau, neurofilament light, Aβ40, and a numerical, non-significant increase in Aβ42 of similar effect size, while p-tau remained largely unchanged, resulting in a significantly decreased p-tau/total tau ratio. |
Notable past biotech efforts
resTORbio
resTORbio is a useful case study in how promising geroscience biology can fail in late-stage development when trial design, endpoint selection, and commercial incentives become misaligned. The company was formed in 2016 after PureTech Health licensed two Phase II–ready mTORC1-targeting assets from Novartis (RTB101 plus an inhaled companion), with the stated goal of treating age-related decline in immune function and potentially expanding into other age-associated diseases. Early data from Novartis’ program helped set the narrative: a short Phase 2a study in adults over 65 reported meaningful reductions in laboratory-confirmed infections and supported the idea that partial mTORC1 inhibition could improve antiviral immunity in older humans. Backed by PureTech’s initial investment and strong investor enthusiasm, resTORbio moved rapidly: after a Series B raise, the company went public early with a valuation that reflected high expectations that a pivotal respiratory trial could establish the first broadly “anti-immunosenescence” therapeutic indication.
The turning point came during the transition from Phase 2b to Phase 3. In its multicenter, randomized, double-blind Phase 2b trial conducted over a winter period (16 weeks treatment plus follow-up), the company reported positive topline findings for RTB101 10 mg once daily, including a reduction in respiratory tract infections that was treated as sufficient justification to progress into a single large registrational study. However, the Phase 2b dataset also raised concerns: the apparent efficacy signal relied heavily on pooling and subgroup decisions, while higher dosing (and some combination regimens) appeared to increase respiratory infections, indicating a narrow therapeutic window and reinforcing that dose selection was not trivial. Despite these issues, resTORbio advanced into PROTECTOR-1, a Phase 3 trial involving> 1,000 participants, selecting RTB101 as the lead agent rather than maintaining the broader combination strategy that had generated the initial signal. This choice made the program more commercially defensible (because RTB101 was proprietary), but also concentrated the clinical risk into a single compound without the support of a better-established comparator.
Phase 3 ultimately failed to meet its primary endpoint, and the reasons illustrate how sensitive geroscience-style trials can be to operational decisions. Near the end of the Phase 3 planning and discussions, the FDA pushed the program away from a virologic, lab-confirmed endpoint and toward a patient-reported symptomatic composite endpoint (“clinically symptomatic respiratory illness”). At the same time, additional major changes were introduced: the Phase 3 population was healthier and more restricted than in Phase 2b (e.g., excluding smokers and certain chronic lung disease groups that had been included previously), and symptom capture shifted from frequent phone-based assessment to daily eDiary self-reporting. These shifts increased measurement noise and made the endpoint more vulnerable to inconsistent reporting and mild symptoms that may not correspond to clinically meaningful infections. In the final analysis, the symptomatic illness endpoint showed essentially no separation between RTB101 and placebo. Importantly, the program did not completely collapse biologically: across Phase 2b and Phase 3, treatment continued to show upregulation of interferon-stimulated antiviral gene programs, suggesting pathway engagement, yet these molecular effects did not translate into a robust, regulator-acceptable clinical benefit under the Phase 3 conditions.
The aftermath was swift and severe. After the Phase 3 failure was announced in November 2019, the company halted development for the respiratory indication and lost market confidence rapidly. Although resTORbio attempted to continue through smaller follow-on programs (including exploratory work in Parkinson’s disease), the lack of a validated clinical success, combined with the cost of running additional large trials without a major pharmaceutical partner, made recovery difficult. The company eventually concluded with a reverse merger into another biotech. In hindsight, resTORbio’s trajectory highlights several practical lessons for longevity translation: small or fragile Phase 2 signals should ideally be reinforced with an intermediate step (or replicated under closely matched conditions), pivotal endpoints must be both clinically meaningful and operationally robust, and changing multiple variables between phases can erase interpretability.
The Mannick 2021 resTORbio program stands out today as one of the most ambitious mTORC1-focused efforts aimed at regulatory approval of a rapalog for an ageing-related condition.
| Name of first author and year of publication | Title of the study | Trial Phase | Number of participants and demographic | Drug/Intervention | Treatment Duration | Primary Outcomes | Key Findings & Safety Results |
|---|---|---|---|---|---|---|---|
| Mannick et al. 2021 [13] | Targeting the biology of ageing with mTOR inhibitors to improve immune function in older adults: phase 2b and phase 3 randomised trials | Phase 2b | 652 participants; adults aged 65–85 years at high risk of RTI (asthma, type 2 diabetes, COPD, congestive heart failure, current smokers, or RTI hospitalisation). | Oral RTB101 (5 mg or 10 mg once daily, or 10 mg twice daily), or RTB101 10 mg plus everolimus 0.1 mg once daily. | 16 weeks | Incidence of laboratory-confirmed respiratory tract infections (RTIs). | RTB101 10 mg once daily significantly reduced laboratory-confirmed RTIs (19% vs 28% placebo). 50% reduction in laboratory-confirmed RTIs with severe symptoms; greatest effect in those >85 years or with asthma. Safe and well-tolerated. |
| Mannick et al. 2021 [13] | Targeting the biology of ageing with mTOR inhibitors to improve immune function in older adults: phase 2b and phase 3 randomised trials | Phase 3 | 1024 participants; adults aged at least 65 years at lower risk of RTI (excluded COPD and current smokers). | Oral RTB101 10 mg once daily or placebo. | 16 weeks | Incidence of clinically symptomatic respiratory illness (symptom-based RTI irrespective of laboratory confirmation). | RTB101 did not reduce the proportion of patients with clinically symptomatic respiratory illness (26% vs 25% placebo). RTB101 increased expression of IFN-responsive genes in whole blood. Treatment was safe and well-tolerated. |
Navitor Pharmaceutical
Navitor Pharmaceuticals represents a branch of “mTOR translation” in industry, built less around chronic mTORC1 inhibition for aging and more around activation of mTORC1 for defined indications, particularly in the CNS.
The company’s flagship program, NV-5138 (also referred to as SPN-820 by its partner), was an orally bioavailable Sestrin2-binding leucine analog designed to transiently activate mTORC1 in the brain, addressing the practical limitation that dietary leucine is rapidly metabolised and does not reliably engage brain mTORC1.
That strategy translated into a partnership with Supernus in 2020 to develop NV-5138/SPN-820 clinically in neuropsychiatric indications, culminating in a larger Phase 2b trial in treatment-resistant depression that reported topline results in February 2025. In that study, SPN-820 did not meet its primary endpoint, despite the sound mechanism and smaller exploratory signals. The value of the Navitor case is not that it validates or invalidates mTOR as a target, but that it illustrates how difficult it is to convert compelling pathway biology into robust clinical benefit when endpoints are noisy and effect sizes may be modest.
At the same time, Navitor’s broader corporate trajectory shows how mTORC1 assets can still create value through indication choice and partnering: in 2022, Janssen acquired Navitor’s spinout Anakuria Therapeutics, built around an mTOR-pathway program aimed at autosomal dominant polycystic kidney disease.
Ongoing/Future Trials
Thanks to growing evidence and significant philanthropic funding, there is now a surge of new clinical trials testing mTOR-based interventions for longevity. These trials are more ambitious in scope: they involve larger sample sizes, longer durations, multi-omics analyses, and creative endpoints. Many are directly informed by the lessons of earlier studies. Below, we highlight several ongoing or upcoming trials of which PI we have interviewed to obtain the most up-to-date information and exclusive insights on how the trials are going.
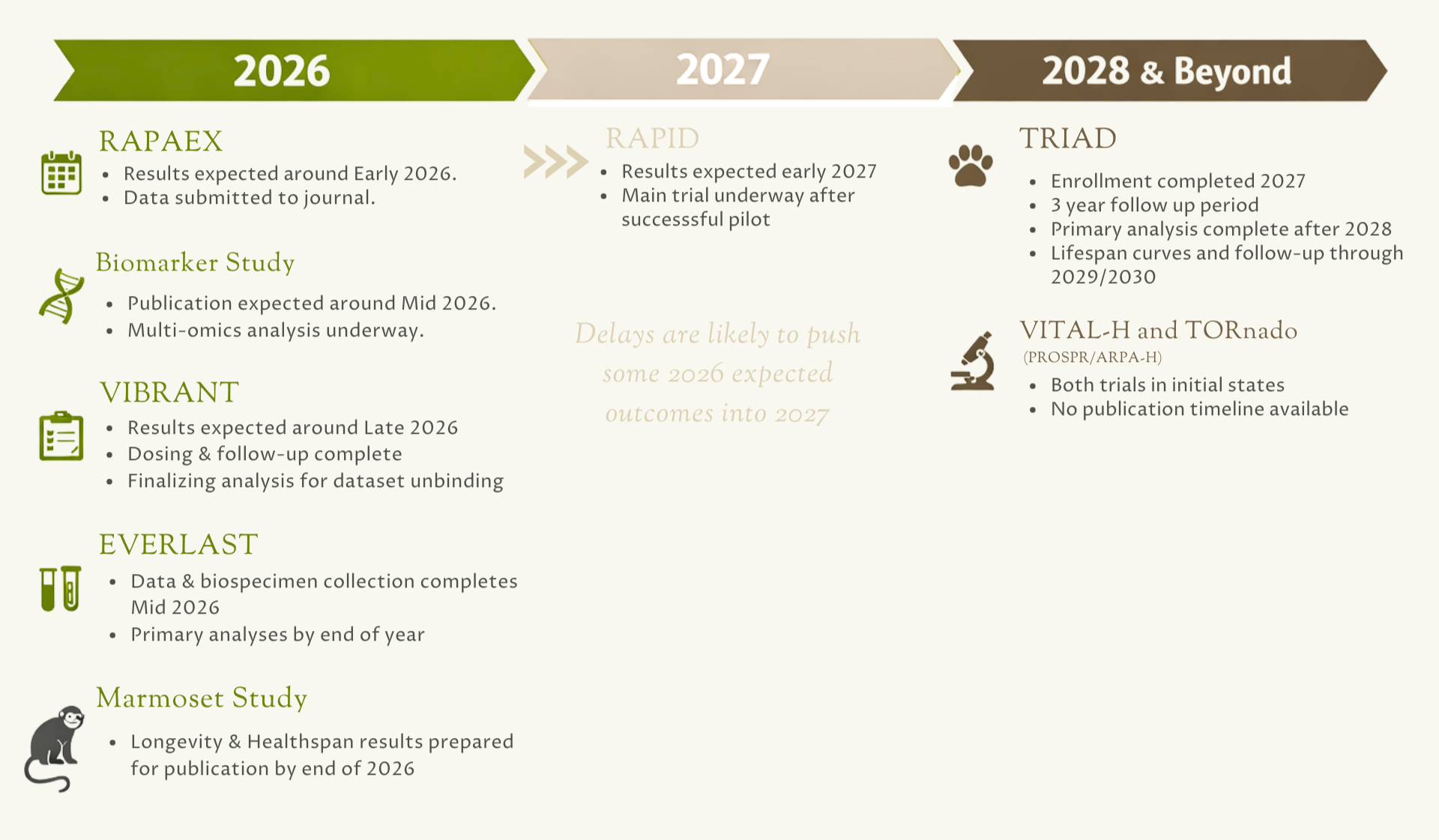
RAPID trial (University of Washington)
Chronic periodontitis (gum disease) is an age-associated inflammatory condition that not only causes tooth loss but has systemic links to health (it’s associated with higher risk of cardiovascular disease and frailty). Dr. Jonathan An and colleagues are launching the RAPID trial to test whether rapamycin can reduce oral “inflammaging” and improve periodontal health, motivated in part by earlier pre-clinical work suggesting rapamycin can improve oral health and improve periodontal disease
RAPID is designed as a double-blind, placebo-controlled study intentionally structured as a foundational, first-in-field study to enable subsequent larger-scale trials. Two major bottlenecks shaped the design and the timeline of the study – regulatory approval and academic infrastructure. From a regulatory standpoint, FDA approval required approximately one year. Because systemic rapamycin had not previously been evaluated for an oral health indication, the Division of Dermatology and Dentistry (FDA) lacked precedent and initially raised safety concerns for use in older adults, requesting IND-backed safety data specific to this population. Few published rapamycin-in-aging studies were IND-enabled in a manner that could be directly leveraged for a dental indication, further slowing the approval process. In parallel, substantial academic infrastructure had to be built de novo. This included establishing compliant electronic medical/dental record workflows, secure data servers, and coordinated data capture across the medical and dental schools, which operate on distinct EMR systems and governance structures. Additional effort was required for ongoing IND maintenance, safety reporting, and cross-school coordination, components that are rarely centralized for dental clinical trials within academic settings.
To address these bottlenecks, the team first conducted a small pilot study (n≈15) to establish safety and feasibility at very low dosages (0.5mg/week and 1mg/week) with careful monitoring of systemic biomarkers, gingival crevicular fluid, and periodontal clinical measures, and innate and adaptive immune responses assessed through PBMCs over a 1-year period (each participant was in the trial for up to 1 year). Systemic and local markers remained stable, while periodontal clinical measures shifted in a direction consistent with the study hypothesis. Importantly, this pilot also served as a full end-to-end validation of the newly built regulatory and academic infrastructure. With this infrastructure now tested in practice, the platform can support future dental, medical clinical trials seamlessly without rebuilding foundational systems.
Participants will be randomized to placebo or one of two low-dose sirolimus regimens administered over an 8-week period, followed by standardized cleaning visits with longitudinal data collection over 9 months. RAPID is positioned not as an endpoint, but as a platform to generate larger, more robust, and rigorous efficacy data in fully powered follow-on trials. Building directly on these findings, the team has submitted a large-scale NIH grant proposal to support a comprehensive, definitive efficacy trial informed by RAPID’s safety, feasibility, and preliminary outcome data.
Biomarker Study (University of Wisconsin)
Dudley Lamming is running a biomarker-centric study to evaluate how selective mTORC1 inhibition maps onto measurable hallmarks of ageing, with the explicit goal of identifying which proposed ageing biomarkers are actually responsive to mTORC1 inhibition and therefore useful for designing geroscience-guided Phase 3 trials. The study is collecting blood from roughly 50 self-reported rapamycin users, mostly off-label users and biohackers, who typically take about 6 mg once weekly and have used it anywhere from one month to three years. To make this feasible, mobile phlebotomy operation is used for drawing blood in participants’ homes across the US, and the team has emphasized that this logistics layer, more than the biology, has been the main operational challenge. The samples are being profiled using multi-omics approaches, including metabolomics and lipidomics; the data are already generated but still undergoing full analysis. The early signals they are comfortable sharing include a decrease in hsCRP, consistent with lower systemic inflammation, and an absence of hyperlipidemia despite this being a commonly cited side effect of rapamycin in other contexts. The investigators plan to publish the results early in 2026, and they explicitly note that Impetus funding was instrumental in getting the work off the ground.
VIBRANT Study (Columbia University)
Co-led by Dr. Yousin Suh and Dr. Zev Williams, this is a prospective, randomized, double-blind, placebo-controlled trial testing whether once-weekly, low-dose oral rapamycin (sirolimus; Rapamune) can slow ovarian aging and delay menopause, motivated by the idea that postponing menopause could reduce downstream risks tied to this transition, including cardiovascular disease, dementia, and increased mortality. The study focuses on ovarian aging because the ovaries appear to age faster than other organs in women, with a steep functional decline beginning around ages 30–35 and culminating near age 50, and the team’s multi-omics comparisons across tissues in younger versus older women suggest mTOR is unusually active in the ovary, consistent with strong preclinical evidence that mTOR inhibition can preserve fertility and ovarian reserve. VIBRANT enrolls women in their 30s and uses ovarian reserve as the primary endpoint, assessed through vaginal ultrasound follicle counts together with hormone measures such as FSH and AMH, while also collecting blood for multi-omics profiling. The original VIBRANT-1 design (6 mg/week for 6 months with follow-up) was revised after substantial institutional and regulatory scrutiny to 5 mg/week for 3 months with 9 months of follow-up, and a second practical bottleneck was finding a pharmacy able to manufacture a placebo that closely matched the active pill to maintain blinding. The trial enrolled 50 participants (25 placebo, 25 treatment) who came to the New York clinic weekly for dosing and were monitored monthly; dosing and follow-up are now complete, and while the team still needs to finalize analysis and unblind the dataset, they report early separation in the primary ovarian-reserve trend lines, no disruption of menstrual cyclicity, and an absence of commonly expected side effects, with results anticipated early in 2026.
The team has also announced the submission of a grant proposal for VIBRANT 2, a follow-up study which will involve 8 institutions across the US with the goal of increasing the sample size and extending the treatment duration to better discern clinically meaningful changes.
RAPAEX
A practical translational concern is whether mTORC1 inhibition interferes with muscle adaptation, especially in older adults using resistance training to preserve function. RAPAEX was conceived in 2021 by Dr. Brad Stanfield after he recognized conflicting signals in the human literature: on one hand, acute mechanistic studies indicate that a single rapamycin exposure around exercise can blunt canonical post-exercise anabolic signaling and muscle protein synthesis, raising concern that mTORC1 inhibition might impair training adaptations if timed inappropriately. On the other hand, older adults, particularly those who are sedentary and at risk of sarcopenia, often have hyperactive mTOR1 signaling at the muscle. Stanfield’s working hypothesis was that weekly rapamycin properly timed might “rebalance” muscle mTORC1 activity in older individuals ultimately aiding the anabolic stimulus of training. To test this directly, RAPAEX randomized two groups initially planned as 20 participants per arm (40 total; 9 dropped out over the study) of previously inactive older adults aged 65–85 into a double-blind, placebo-controlled design where both arms completed an exercise program three days per week on Monday, Wednesday, and Friday, while receiving either placebo or sirolimus 6 mg once per week on Saturdays for 13 weeks. The 30-second chair stand test was the primary endpoint, with grip strength, 6-minute walk test, bloodwork, and the SF-36 wellbeing questionnaire as secondary endpoints. A major practical issue was producing a placebo that matched the intervention capsules; the team solved this with a simple but effective blinding strategy by using standard Pfizer sirolimus tablets placed inside a larger white opaque capsule filled with cellulose, while the placebo group received the identical larger capsule filled with cellulose but without the sirolimus tablet inside, and no participant identified this workaround during the trial. From an execution standpoint, Stanfield emphasized that the largest hurdle was funding the study, and that operational success depended heavily on providing clear, detailed instructions to the clinical and hospital staff delivering the protocol to minimize dropouts and other avoidable logistical problems. The study has now concluded, with data collected, analyzed, and submitted to a journal.
Additional Notable Trials
EVERLAST (University of Wisconsin, Madison)
EVERLAST (Everolimus Aging Study) is a Phase 2, randomized, double-blind, placebo-controlled trial designed to test whether pharmacologic mTORC1 inhibition produces measurable changes in aging-relevant physiology and candidate biomarkers in a metabolically enriched older population. The trial has completed enrollment and reached its target sample size of 72 insulin-resistant adults aged 55–80, a design choice that prioritizes clinical detectability by studying individuals with mild-to-moderate metabolic dysfunction rather than exceptionally healthy participants. EVERLAST is funded primarily by the NIH/NIA, with Impetus Grants supporting additional biomarker-focused analyses intended to quantify molecular signatures that may shift under treatment. The investigators anticipate completing data and biospecimen collection in July 2026, after which the primary analyses can be finalized.
RAP PAC (University of Wisconsin, Madison)
In parallel, RAP PAC is an NIH/NIA-funded Phase 1 dose-identification study that is currently ongoing and is intended to map tolerable and biologically active dosing regimens for rapamycin and everolimus in humans.
REACH (University of Texas, San Antonio)
REACH is a Phase 2, randomized, placebo-controlled clinical trial evaluating the safety, tolerability, and feasibility of 12 months of oral rapamycin in older adults with amnestic mild cognitive impairment (aMCI) and early-stage Alzheimer’s disease. The primary objective emphasizes feasibility and safety, but the trial is also positioned to generate outcome data including cognitive measures and Alzheimer’s-related biomarkers.
Marmoset Trial (University of Texas, San Antonio)
It’s also worth highlighting marmoset trial led by Adam Salmon at the University of Texas, which tests rapamycin in common marmosets, a new world monkey species, in a controlled trial. The rationale is that marmosets recapitulate several features of human aging more closely than mice, including a broader spectrum of age-related morbidity, whereas laboratory mice often die predominantly from cancer or strain-specific pathology. Nonhuman primates share many of the diseases of age with humans, and thus might inform more on aging effects of rapamycin vs disease specific effects. To address this, his group initiated a long-duration study nearly ten years ago in 66 marmosets split evenly between treatment and control arms. The trial enrolled older animals at baseline (average age ~7 years, with an average lifespan of ~10 years) sourced from multiple facilities across the United States, creating a heterogeneous cohort with diverse backgrounds that better resembles real-world human variability. The intervention uses daily rapamycin at 1 mg/kg, a regimen comparable to doses used in mouse lifespan studies and intended to achieve blood concentrations similar to those seen in transplant medicine, substantially higher than the intermittent, low-dose schedules commonly used off-label for longevity in humans. The primary endpoint is survival, with longitudinal monitoring that includes safety markers, cognitive testing, and selected functional measures; the team also collected blood for multi-omics profiling and for pharmacodynamic assessment of target engagement in circulating cells via S6K phosphorylation, and performed systematic tissue collection at death for pathological evaluation, which remains ongoing. Previous reports from this study have shown that rapamycin was well-tolerated without clear serious side effects in this group, and that mTOR signaling was inhibited though with inter-individual variability. The effects of rapamycin on longevity and healthy aging in this cohort are being prepared for publication in the near future.
TRIAD
Finally, another bridge study is represented by TRIAD, the large, multicenter, randomized rapamycin trial in companion dogs led by Matt Kaeberlein. This isn't a human trial, but it is one of the most informative translation layers between mice and humans because pet dogs share environmental exposures, spontaneous disease patterns, and clinically meaningful outcomes in a compressed timeframe. The trial is explicitly designed to test whether rapamycin improves healthspan metrics and potentially lifespan in middle-aged dogs, using clinical-trial methodology (randomized, double-masked, placebo-controlled, multicenter). The study launched in 2020 but was interrupted by the pandemic and subsequently faced an 18-month funding gap before securing new support and restarting. The trial is statistically powered to enroll around 600 dogs and detect a 9% change in lifespan over a three-year follow-up period that follows a one-year treatment period during which dogs receive low-dose weekly rapamycin. Enrollment is expected to complete within the next year, after which all enrolled dogs will be followed for three years before a formal primary analysis is conducted. Importantly, this analysis will not yet include lifespan data — the team does not expect to publish survival curves before 2029. Around 200 dogs are currently in some stage of the study, either having completed treatment and follow-up, actively receiving treatment, or in the follow-up phase. The dataset remains blinded, and no interim signal has prompted early unmasking; no major side effects have been detected. One possible complication is that the team is not observing the rate of deaths they originally anticipated, likely due to a healthy cohort bias. Because enrollment selected for otherwise healthy dogs rather than a broadly representative population, the baseline mortality rate may be lower than the power calculation assumed, potentially requiring extended follow-up beyond the initial three years to accumulate sufficient events. TRIAD is among the most anticipated studies in the field: its sample size, duration, and clinical relevance mean that a positive result would carry more weight than almost anything else currently in progress.
VITAL-H and TORnado (ARPA-H PROSPR Program)
In early 2026, the federal government placed a significant bet on the longevity field. ARPA-H, the health-focused cousin of DARPA, created to fund high-risk, high-reward biomedical research, announced the winners of its PROSPR program, distributing up to $144 million across seven teams with the explicit goal of treating aging as something that can be slowed, or even reversed. Two of those seven awards landed in mTOR territory — a meaningful concentration in a single pathway, and a reflection of how central mTORC1 inhibition has become to the field’s translational ambitions in 2026.
The Barshop Institute at UT Health San Antonio received up to $38 million to run VITAL-H (Validation and Intervention Testing for Aging, Longevity and Healthspan), which is positioned as the first Phase 3 clinical trial targeting ageing in adults not yet diagnosed with a specific disease, randomising generally healthy individuals aged 60–65 to rapamycin, dapagliflozin, or semaglutide. The trial’s use of a composite “Intrinsic Capacity” endpoint spanning cognition, mobility, psychological well-being, vitality, and sensory function reflects a deliberate attempt to operationalise ageing as a measurable, multidimensional construct rather than a single disease.
Cambrian Biopharma’s PROSPR award of up to $30.8 million takes a different angle. Rather than working with rapamycin itself, their TORnado platform has produced a new class of compounds designed to hit mTORC1 selectively, without mTORC2 engagement that many researchers believe is responsible for rapamycin’s less desirable metabolic effects.
Timeline of Expected Trial Outcomes - Produced by Norn Group
Notable Current Biotech Efforts
Aeovian Pharmaceuticals
Aeovian Pharmaceuticals is an instructive example of a different translational strategy for mTORC1 biology than the “broad ageing indication” route attempted by some earlier programs. Rather than starting in heterogeneous older populations where endpoints are difficult and signal-to-noise is poor, Aeovian has prioritized genetically defined diseases where mTORC1 hyperactivation is causal, with the goal of de-risking mechanism, dose selection, and target engagement before expanding into broader chronic and age-associated indications. The company was originally incubated at the Buck Institute for Research on Aging and has described itself as building a pipeline of highly selective mTORC1 inhibitors designed to restore “metabolic quality control” while avoiding the tolerability constraints that can occur when less selective mTOR inhibitors impact other nodes of the pathway. This positioning is central to their thesis: in mTOR-driven disorders such as tuberous sclerosis complex (TSC), mTORC1 signaling is pathologically elevated due to loss-of-function mutations in TSC1/TSC2, and existing treatment approaches (including mTOR inhibitors used in related settings) can be limited by adverse effects that restrict chronic use and dose intensity. Aeovian’s lead program is therefore framed as a pharmacology problem as much as a biology problem, building an inhibitor with strong mTORC1 selectivity plus adequate CNS penetration, since epilepsy is among the most common and clinically burdensome manifestations in TSC.
This approach has translated into a clinical development plan centered on AV078, which Aeovian describes as a first-in-class, CNS-penetrant selective mTORC1 inhibitor intended for refractory epilepsy associated with TSC. In March 2024, the company announced dosing of the first cohort in a Phase 1 clinical trial and raised an additional $50 million to support completion of early clinical development and preparation for subsequent Phase 2 testing. Public statements around the 2024 financing repeatedly emphasize two priorities: (i) establishing safety and pharmacology in healthy volunteers and (ii) moving into adult and pediatric TSC epilepsy populations where the clinical endpoints and patient need are clear. In December 2025, Aeovian disclosed a further $55 million Series B to advance AV078 into Phase 2 development in TSC-related refractory epilepsy, again highlighting the intent to deliver improved efficacy and tolerability compared with current options and to achieve sustained seizure control in patients whose seizures remain inadequately controlled. Across these announcements, the company’s messaging is consistent: prioritize a setting where mTORC1 is clearly the driver, where CNS exposure matters, and where efficacy can be read out with established neurological endpoints.
From a translational perspective, Aeovian’s program addresses several limitations that have complicated mTORC1 development in broader aging trials. First, rare genetic disorders such as TSC represent a “high-mechanism-confidence” clinical context: pathway hyperactivation is present by definition, reducing uncertainty about whether an mTORC1 inhibitor is addressing the causal biology. Second, epilepsy provides clinically validated outcomes (e.g., seizure frequency and severity) that are more interpretable than composite symptom endpoints used in respiratory or general healthspan studies. Third, Aeovian’s emphasis on selective mTORC1 inhibition reflects a core hypothesis in the field: that improved selectivity may allow chronic pathway modulation with fewer dose-limiting tradeoffs, which is likely to be essential for any long-term indication. Finally, the company has explicitly positioned AV078 as a platform entry point rather than a single product, arguing that once an mTORC1-selective CNS-active inhibitor is clinically validated in a high-signal disease, the same pharmacology could be explored in other mTORC1-mediated neurological and age-associated conditions.
Gaps and Future Direction
Despite the progress, several critical gaps remain in our knowledge and approach. Addressing these will be vital for the success of mTOR-based longevity therapies and ageing trials in general:
Lack of Validated Surrogate and Biomarkers
Aging trials currently lack an accepted surrogate marker that reliably predicts clinical benefit. Unlike blood pressure for cardiovascular disease or HbA1c for diabetes, we don’t have a single metric for “biological aging.” Epigenetic “aging clocks,” proteomic or metabolomic signatures, and functional indices (like gait speed or grip strength) are all candidates but none are yet proven surrogates. Moreover, many studies failed to measure whether mTOR was actually inhibited. We assume a dose had an effect, but we don’t know if some participants were non-compliant or hyper-metabolizers. What is the best easily accessible biomarker of mTORC1 activity in humans? Muscle biopsies can show phospho-S6 reduction, but those aren’t always available. Blood immune cells are an alternative but phosphorylations are extremely labile and hard to reliably measure in humans. In our interview, Joan Mannick highlighted (and Adam Salmon from the Marmoset study confirmed) how there is a huge variability in baseline mTORC1 activation between individuals and how we don’t really have a way to stratify participants by mTORC1 activation and a way to gauge target engagement via the drug.
These limitations are increasingly recognized in the clinical development community focused on geroscience. A recent Norn Group essay argues that, because validated ageing biomarkers are not yet strong enough for pivotal decision-making, the most feasible path for first-in-class “aging trials” is to rely on reimbursable clinical outcomes rather than clocks alone. Specifically, the authors propose a multi-morbidity composite endpoint, counting time-to-first occurrence across several major age-related events, as a practical way to increase event rates and reduce sample size while staying aligned with outcomes that regulators and payers already accept. This matches the direction taken by composite endpoint designs and provides a concrete rationale for why trials like TAME (Targeting Aging with Metformin) and VITAL-H use multimorbidity endpoints instead of single biomarkers.
Endpoints and Goals
Academic studies tend to cast a wide net on endpoints, for example, measuring dozens of biomarkers and multiple functional outcomes to see where an intervention has an effect. The downside is academics sometimes lack a single strong primary outcome, making it harder to declare “success” in a traditional sense. This is a major concern for the translatability of academic findings, as the FDA cares about a drug only if it improves how a patient feels, functions or survives. Biotechs, in contrast, zero in on one clinically recognized outcome as primary endpoint. This focus is necessary for regulatory approval and investment purposes, but it can be a double-edged sword. As we saw with resTORbio, picking the wrong primary endpoint (or one that’s too ambitious) can sink a trial even if the drug has real biological activity. As mentioned, aging itself isn’t an approved indication. To convince regulators, we may need to do what TAME and VITAL-H are attempting: propose a composite endpoint that essentially captures “aging” (e.g. any of several age-related diseases/events). The FDA has shown some openness but will require strong evidence. Ethically, exposing healthy people to a drug with any risk requires clear rationale.
Optimal Dosing Regimen Unclear
There is still no consensus on how to dose rapamycin for aging. Is it better to give a moderate dose weekly? A higher dose biweekly? Short pulses (a few months on, then off)? The “goldilocks” regimen that maximizes mTORC1 inhibition while minimizing mTORC2 effects is hypothesized but not confirmed. Perhaps different regimens work for different tissues (e.g. immune system might need shorter, frequent dosing whereas metabolic tissues might prefer intermittent). We might consider moving toward a personalized dosing: monitoring biomarkers in each patient and adjusting accordingly (for example, if a patient’s phospho-S6 is still high on current dose, escalate it; if their lipids spike too much, reduce frequency).
Long-term Safety in Healthy Individuals
Long-term safety is one of the main constraints that affects the adoption of mTORC1 inhibitors. While multiple studies support short-term tolerability over weeks to months, there is still limited evidence on multi-year exposure in otherwise healthy older adults, which is the relevant scenario for any preventive aging strategy. Because mTORC1 controls core anabolic and repair programs, systemic inhibition can produce adverse events that are acceptable in high-need disease settings but less acceptable in generally healthy participants. This creates a higher evidentiary burden for trial design, monitoring, and endpoint selection, and helps explain why broad “aging trials” have been more readily proposed for safer, widely used repurposed agents, such as metformin, whereas rapamycin-class drugs have progressed primarily through disease-anchored studies and/or with conservative dosing strategies. Classic high-dose immunosuppression is associated with higher rates of infection and metabolic disturbance, and although geroscience trials use lower or intermittent dosing, rare or cumulative harms may only become detectable over years. In addition, some potential liabilities may not present as acute adverse events but as slowly accumulating deficits in tissue maintenance; for example, rodent studies have reported cataracts and degeneration of thymic or gonadal tissues under lifelong rapamycin exposure in some strains, raising the possibility that long-duration monitoring in humans will be necessary even when short-term studies appear benign. Early observational comparisons of off-label rapamycin users versus non-users have been broadly reassuring over roughly two years, with few differences in self-reported health aside from increased mouth ulcers, but these datasets remain limited by self-selection and reporting bias. For these reasons, preventive trial designs should either anchor efficacy to clearly meaningful clinical outcomes or focus on high-risk subgroups where the benefit-to-risk ratio is more favorable, while building in long-term safety follow-up data.
Conclusion
mTOR inhibition remains one of the most promising and extensively validated strategies in geroscience for slowing biological aging and preventing age-related disease. More than 14 years have passed since rapamycin first extended lifespan in mice, and in that time the field has moved from largely preclinical promise to a growing portfolio of human trials. These studies have shown that mTOR biology can be modulated safely enough to test in humans, and that intervention can produce measurable effects across multiple aging-relevant domains.
At the same time, the evidence reviewed here makes clear that biological plausibility is not enough. The outcomes of mTORC1-targeting trials depend heavily on indication selection, dose and schedule, endpoint design, target-engagement measurement, and operational execution. In other words, the central challenge is no longer only whether mTOR is relevant to aging, but how to test that biology in humans in a way that yields interpretable and clinically meaningful evidence.
The field has, nevertheless, made real progress. Past studies have been essential for establishing feasibility, exploring biomarker panels, and identifying potential signal areas, while ongoing trials are increasingly adopting disease-anchored indications and clearer outcome measures. Important uncertainties remain, especially around long-term safety in healthy individuals, validated surrogate biomarkers of aging, and the optimal strategy for selective mTORC1 modulation.
Taken together, the current evidence supports the conclusion that mTORC1 inhibition is not yet a proven clinical longevity intervention, but it remains one of the most credible entry points for building one. The next phase will depend on rigorous trial design, long-term safety characterization, and reproducible effects across populations and endpoints. If those conditions are met, mTORC1-targeting drugs could become the first inarguable longevity therapies.
This piece was produced as a Talent Bridge Award Project.
To learn more about how you can support Talent Bridge and Norn Group click the button below.
-
Roark KM & Iffland PH II (2025). Rapamycin for longevity: the pros, the cons, and future perspectives. Frontiers in Aging, 6: 1628187. DOI: 10.3389/fragi.2025.1628187.
Konopka AR & Lamming DW (2023). Blazing a trail for the clinical use of rapamycin as a geroprotecTOR. GeroScience, 45(5): 2769–2783. DOI: 10.1007/s11357-023-00935-x.
Lee DJW, Hodzic Kuerec A & Maier AB (2024). Targeting ageing with rapamycin and its derivatives in humans: a systematic review. The Lancet Healthy Longevity, 5(2): e152–e162. DOI: 10.1016/S2666-7568(23)00258-1.
Harrison DE, Strong R, Sharp ZD, Nelson JF, Astle CM, Flurkey K, Nadon NL, Wilkinson JE, Frenkel K, Carter CS, Pahor M, Javors MA, Fernandez E, Miller RA, et al. (2009). Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature, 460: 392–395. DOI: 10.1038/nature08221.
Mannick JB, Del Giudice G, Lattanzi M, Valiante NM, Praestgaard J, Huang B, Lonetto MA, Maecker HT, Kovarik J, Carson S, Glass DJ, Klickstein LB (2014). mTOR inhibition improves immune function in the elderly. Science Translational Medicine, 6(268): 268ra179. DOI: 10.1126/scitranslmed.3009892.
Mannick JB, Morris M, Hockey H-UP, Roma G, Beibel M, Kulmatycki K, Watkins M, Shavlakadze T, Zhou W, Quinn D, Glass DJ, Klickstein LB (2018). TORC1 inhibition enhances immune function and reduces infections in the elderly. Science Translational Medicine, 10(449): eaaq1564. DOI: 10.1126/scitranslmed.aaq1564.
Kraig E, Linehan LA, Liang H, Romo TQ, Liu Q, Wu Y, Benavides AD, Curiel TJ, Javors MA, Musi N, Chiodo L, Koek W, Gelfond JAL, Kellogg DL Jr (2018). A randomized control trial to establish the feasibility and safety of rapamycin treatment in an older human cohort: Immunological, physical performance, and cognitive effects. Experimental Gerontology, 105: 53–69. DOI: 10.1016/j.exger.2017.12.026.
Chung CL, Lawrence I, Hoffman M, Elgindi D, Nadhan K, Potnis M, Jin A, Sershon C, Binnebose R, Lorenzini A, Sell C (2019). Topical rapamycin reduces markers of senescence and aging in human skin: an exploratory, prospective, randomized trial. GeroScience, 41(6): 861–869. DOI: 10.1007/s11357-019-00113-y.
Gonzales MM, Garbarino VR, Kautz TF, Song X, Lopez-Cruzan M, Linehan L, Van Skike CE, De Erausquin GA, Galvan V, Orr ME, Musi N, He Y, Bateman RJ, Wang C-P, Seshadri S, Kraig E, Kellogg D Jr (2025). Rapamycin treatment for Alzheimer’s disease and related dementias: a pilot phase 1 clinical trial. Communications Medicine, 5(1): 189. DOI: 10.1038/s43856-025-00904-9.
Moel M, Harinath G, Lee V, Nyquist A, Morgan SL, Isman A, Zalzala S (2025). Influence of rapamycin on safety and healthspan metrics after one year: PEARL trial results. Aging (Albany NY), 17(4): 908–936. DOI: 10.18632/aging.206235.
Moody AJ, Wu Y, Romo TQ, Koek W, Li J, Linehan LA, Clarke GD, Yang EY, Espinoza SE, Musi N, Chilton RJ, Kraig E, Kellogg DL Jr (2025). Short-term mTOR inhibition by rapamycin improves cardiac and endothelial function in older men: a proof-of concept pilot study. GeroScience. 2025 Sep 19 (Online ahead of print). DOI: 10.1007/s11357-025-01855-8.
Svensson JE, Dörfel RP, Schain M, Bolin M, Sacuiu S, Hagman G, Forsberg Morén A, Kivipelto M, Plavén-Sigray P (2025). Evaluation of rapamycin as a neuroprotective treatment in Alzheimer’s disease: a six-month single-arm open-label clinical pilot trial. medRxiv (Preprint). DOI: 10.64898/2025.12.08.25340853.
Mannick JB, Teo G, Bernardo P, Quinn D, Russell K, Klickstein L, Marshall W, Shergill S (2021). Targeting the biology of ageing with mTOR inhibitors to improve immune function in older adults: phase 2b and phase 3 randomised trials. The Lancet Healthy Longevity, 2(5): e250–e262. DOI: 10.1016/S2666-7568(21)00062-3.
-
Thanks to Soleil Wizman (Norn Group) for her help shaping the resTORbio section and the principal investigators who generously dedicated time for exclusive interviews and clarifications that informed this review: Jonathan An, Yousin Suh, Dudley Lamming, Brad Stanfield, Joan Mannick, Adam Salmon, and Matt Kaeberlein.
Published March 2026.
© Norn Group 2026